
The limonoids are extremely important secondary metabolites in citrus fruit because of their impact on bitterness in juices. This class of compounds are all highly oxygenated triterpenes and contain a furan ring attached to the D-ring (Hasegawa and Miyake, 1996). Different types of oxidation with skeletal rearrangement produce other types of limonoids (Endo et al., 2002; Hasegawa, 2000).
Excellent reviews exist on the analysis and chemistry of the limonoids (Roy & Saraf, 2006; Manners, 2007; Tundis et al., 2014; Gualdani et al., 2016) The term ‘limonoid’ is derived from limonin (CAS 1180-71-8), which was first identified as the bitter constituent of Citrus seeds in 1841.
The limonoids can be found in leaves, seeds, pulp and juice. It takes two forms: the aglycone which has the bitterness taste and as a glucoside where it is bound to glucose and potentially other types of sugar moieties. At least 37 limonoid aglycones and 17 limonoid glucosides have been isolated from citrus and other members in closely related families (Mandadi et al., 2007). Citrus seed is a significant source industrially of limonoids and forms about 1% w/w of the total seed as fresh weight.
The other types of limonoids besides limonin are nomilin, nomilinic acid, obacunone, deacetylnomilin. They are related to the tetranortriterpenoid compounds such as azadirachtin extracted from the neem tree (Azadirachta indica). This particular compound is an insecticide.
Most important aglycones are low to medium polarity compounds and usually soluble in organic solvents as well as aqueous solutions.
The Content Of Limonoids In Fruit
The seeds are the main sources of limonoids but usually as the aglycone but they can also be found in many other parts of the plant. The peel is another source. The glucosides tend to be concentrated in the juice and pulps.
Analysis of the juices show they can contain 300-330 ppm, 190 ppm and 82 ppm of limonoids in orange, grapefruit and lemon juices respectively. The major glucoside in citrus juices was limonin 17‐O‐β‐D‐glucopyianoside. This constitutes over 50% of the total limonoid glucosides in the juices. (Fong et al., 1989).
The citrus fruit pummelo [Citrus grandis (L.) Osbeck] contains on average 18 ppm limonin and 29 ppm total limonoid glucosides (Ohta & Hasegawa, 1995). A more recent study revealed 11.5 mg/g of seeds could be obtained (Qin et al., 2018) This is high compared to other juices. Pummelo seed contains nomilin, obacunone and trace amounts of deacetylnomilin as well as their equivalent as 17–β‐D‐glucopyranoside derivatives (glucosides). Total limonoid aglycone concentration in the seeds ranged from 773 ppm to 9,900 ppm and total limonoid glucosides ranged from 130 ppm to 1,912 ppm. Bergamot is a source of limonoids (Russo et al., 2016).
As the fruits mature, there is a rapid accumulation of limonoids especially in the seeds.
The essential oil is a good source too:- the content of limonin ranged from 0.5 in a clementine essential oil to 21.2 mg L−1 (bergamot essential oil) which were cold-pressed citrus essential oils (Zoccali et al., 2018)
Limonin
Limonin (7,16-dioxo-7,16-dideoxylimondiol) is a highly oxygenated triterpenoid aglycone which belongs in the class of furanolactones and is extracted from mainly citrus fruits. It is probably the most common limonoid found in citrus fruit. It is a derivative composed of a furan ring and an epoxide ring and is often called a limonoate D-ring-lactone or limonoic acid di-delta-lactone.
Limonin has been known by different names in the past including obaculactone, evodin and dictamnolactone mainly because of the different plant sources from which it has been obtained.
The compound is isolated from various members of the plant families in the Rutaceae (Champagne et al., 1992) and the Meliaceae. The Meliaceae family is a rich source of the limonoid called meliacin (YT, 2011). The limonoids can also be found in the Cneoraceae, Ptaeroxylaceae, and Simaroubaceae families.
The Japanese Bitter Orange (Citrus trifoliata) is a strong source but it may be reclassified in future as a new genus and the plant renamed as Poncirus trifoliata.
Limonin is strongly associated with bitterness of flavour in citrus fruit, especially grapefruit and navel orange but is also found in the Dictamnus genus (Yoon et al., 2010). High quantities occur in both orange, pummelo and lemon seeds. For many consumers, bitterness is highly problematic and its presence often reduces the commercial value of the juice.
The Issue Of Bitterness
Limonin can be highly problematic in fruit juices because in sufficient quantity it produces unacceptable bitterness. Back in the late 1940s it was found that limonin produced a delayed bitterness in citrus fruits. It’s worth stating that any immediate bitterness in a juice is due to naringin but that is not the subject of this article.
Bitterness itself is caused by two types of compounds: the flavonoids which are found in grapefruit, bitter orange and pummelo. The bitterness produced by limonin occurs in these bitter varieties and the non-bitter types too.
Any bitter limonoids in concentrations above 6ppm reduce the acceptability of citrus juices and restricting the commercial sale of the juice. The taste threshold for limonin depends on the person. Some people are sensitive to just 0.5ppm limonin whilst in the general population, the threshold is in the range of between 2 and 6 ppm. For the majority, the most objectionable level is between 8 and 12 ppm limonin in juice. Navel oranges generate a juice in the range 10 to 30ppm whilst sweet oranges do not produce limonin at all or at most 6ppm. The ideal juice should have a maximum specification of between 6 and 7ppm. The level of nomilin in juice is less than 2ppm and has not been truly perceived as an issue in bitterness.
Generally speaking the occurrence of limonin depends on the point in the ripening of fruit. Take the production of orange juice in Israel from the Shamouti variety (Flavian & Levi, 1970). No processing of fruit occurs in the early part of the season because the juice has a highly bitter flavor. Leave it long enough and both precursor and limonin contents fall away to a negligible point.
Purification Of Limonoids
The main method on a laboratory scale is extraction using solvents (Melwita & Ju, 2010; Vikram et al., 2007).
Significant amounts are usually prepared by preparative HPLC or HPLC/PDA/MS from the seeds. At the moment though there is really no satisfactory method for large-scale extraction although some promise was possible using an aqueous system. This was tried on limonin extraction from Citrus reticulate (Liu et al., 2012).
It is possible to convert limonin which is water-insoluble to limonoate A-ring lactone (water-soluble) in an alkaline environment (Fisher, 1978), while limonoate A-ring lactone can be reconverted into limonin in an acidic environment. The reversal as it were at acidic pH occurs via the reversible ring-opening of the limonin D ring at different pHs. This is a useful reversible attribute that has yet to be properly exploited on a bigger scale although there are probably hints that it is being conducted commercially.
The limonoids can also be extracted from the seeds using hydrotropic extraction. This is a process where an agent solubilises a hydrophobic compound in water. the extraction efficiency is dependent on the hydrotrope concentration, extraction temperature and percent of raw material loaded. Two hydrotropes such as sodium salicylate (Na-Sal) and sodium cumene sulphonate (Na-CuS) have been effective for limonoids (Dandekar et al., 2008).
Analysis Of Limonoids
The citrus limonoids are unique for many species and varieties, and make excellent taxonomic markers. This is extremely valuable in the fight against adulteration with poorer quality juices. Thin-layer chromatography has been the method of choice in the past but as mentioned earlier high performance liquid chromatography (HPLC) is now employed (Rousseff & Fisher, 1980).
Limonoids can be characterized in juice as the aglycone (Zoccali et al., 2018) using supercritical fluid chromatography tandem mass spectrometry. More conventional methods use a Fourier transform infrared (FT-IR) spectrometer and nuclear magnetic resonance (NMR) identification.
Biosynthesis Of Limonoids
The limonoids are all synthesized through the terpenoid biosynthetic pathway. There are four different biogenetic pathways identified for the biosynthesis of limonoids in Citrus fruit. Each of the pathways is identified by the major limonoid produced. We have limonin, calamin, ichangin and 7-alpha acetate limonin.
Synthesis always starts with squalene cyclization and proceeds via the cytoplasmic based acetate-mevalonate pathways. Nomilin is the main precursor to all the other limonoids. It is synthesized in the phloem region of the stems and then translocated to other parts of plants such as the leaves, fruit tissues, peels, and seeds where it is further metabolized to other limonoids.
The citrus limonoid aglycones are then glucosidated by limonoid UDP‐D‐glucose transferase in maturing fruit tissues and seeds. The glucosides accumulate to considerable levels in the pulp of the fruit. This is the basis of their significance as a secondary metabolite.
Reduction Of Bitterness
Citrus fruit is notorious for being prone to bitterness especially if the juice is poorly processed, left even for a few hours before further processing or allowed to ferment. Despite the abundance of citrus fruit such as navel oranges for example, very little of this fruit is ever consumed in a form where it is unblended. Navel oranges in particular are prone to bitterness following juice extraction.
Citrus growers realised early on that juice from late-season navel oranges was less bitter than fruit picked much earlier. Unfortunately, the low bitterness levels were only seen in very late-picked fruit when the optimum ripeness for distribution was much earlier. To induce the natural debittering process, growers and researchers tried storing the fruit in warm, moist rooms. prolonged storage did indeed achieve some debittering but the major drawback was growth of molds and bacteria, the time and space needed for such a large-scale process, production of off-flavours and the requirement for specialist storage rooms.
Citrus species contain a precursor of limonin which is the limonoate A-ring lactone. It is tasteless but following juice extraction, the acidic pH encourages the conversion of the limonoate A-ring to its bitter derivative, limonin. The conversion is also accelerated by the action of limonin D-ring lactone hydrolase which is present in citrus juices (Maier et al., 1969).
Its presence is only noted after a few hours when the fruit is pulped and the juice expressed. The delayed onset of bitterness means that the consumer acceptance of either the processed or fresh product is reduced.
Processes to eliminate bitter principles from juice have often focused on the removal of limonin although hesperidin and naringin which are also found in fruit juices have also long been associated with bitterness. These too are often destroyed and degraded to reduce the overall bitterness of the juice.
Some microorganisms i.e. bacteria possess the enzyme limonoate dehydrogenase which prevents the formation of limonin by catalyzing the oxidation of the limonoate A-ring lactone into the corresponding 17-dehydrolimonoate which is a non-bitter derivative instead. Limonin cannot be produced from this molecule.
The bacteria known to possess this enzyme include Arthrobacter globiformis, Pseudomonas sp. Strain 321‐18, Cornyebacterium fascians, Rhodococcus fascians and Azadirachta excelsa. These micro‐organisms are known to metabolize limonin via 17‐dehydrolimonoid and deoxylimonoid pathways.
Successful removal methods often exploit immobilised enzyme and cell technology. For example, proof of principle was shown with Navel orange juice serum. The serum was successfully debittered using A. globiformis cells immobilized in an acrylamide gel. In this instance the serum containing between 10 and 27 ppm limonin was fed into a small column (16ml). Whilst this was a small-lab scale study it gives a pointer as to what is possible. The key fact was the content of limonin was reduced by more than 70% (Hasegawa et al., 1982). Immobilised cells of Corynebacterium fascians immobilized in acrylamide gel also reduce limonin content (Hasegawa et al., 1985).
Adsorbents are also effective at removing limonin and other limonoids but they can scalp other flavor compounds.
Some success could be achieved using polyamide as an adsorbent (Chandler et al., 1968). It probably requires a couple of passes to achieve an acceptable reduction but there is an issue with leakage of the amide group into the juice being debittered if column preparation is poor. It is worth considering cellulose acetate because that shows some selectivity for limonin (Chandler and Johnson, 1977). They claimed to remove between 44 and 70 % of the limonin content in less than an hour but not hesperidin or ascorbic acid.
Grapefruit juice has often been treated this way using materials such as polystyrene divinylbenzene (Manlan et al., 1990). Adsorbents based on these resins have largely been adopted in preference to any other type including activated carbon, cellulose acetate and zeolites.
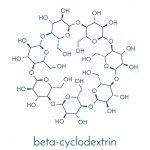
Cyclodextrin is a good ingredient for sequestering certain flavor compounds including limonin and can be bound to columns containing XAD resins which are then operated in a fluidized bed (Wilson III et al., 1989). When the cyclodextrin content in a juice is 0.5% w/w it will reduce the bitterness of both limonin and naringin by half (Konno et al., 1982).
Amberlite IR 120 and IR 400 are useful adsorbents and when coupled with naringinase entrapped in alginate beads, will also reduce grapefruit juice tartness (Mishra et al., 2003).
The addition of neodiosmin makes for a very effective suppressor of limonin bitterness by raising the limonin bitterness threshold in orange juice and so reducing bitterness in juice. The compound is also effective at suppressing naringin bitterness. The use of 50 to 150 ppm is recommended for reducing bitterness and improving palatability and acceptability.
Sucrose is weak suppressor. Citric acid is more pronounced. Other sweeteners such as neohesperidin dihyrdrochalcone, hesperidin dihydrochalcone & aspartylphenylalanine methyl ester have all been shown to reduce bitterness to some extent.
Auxins which are growth hormones like indolacetic acid, indolebutyric A, napthaleneacetic acid and 2,4,5-trichlorophenoxyactic acid are potent inhibitors of nomilin biosynthesis.
Another approach is to grow varieties which produce low levels of limonin precursor before harvesting. Research on creation of transgenic citrus trees that produce fruits free of the limonoid bitterness problem is in progress.
A genetic engineering approach has been to identify specific genes coding for enzymes that produce bitter tasting limonoids. The goal here is to literally block aglycone formation by inserting disrupting pieces of DNA, to divert limonin production down another pathway or to downregulate such genes. Three target enzymes have been identified:-
(1) Nomilin deacetylase – this enzyme is especially active in the citrus fruit C. ichangensis. This citrus species accumulate nonbitter deacetylnomilin and ichangensin instead of limonin in the fruit pulp and seeds. It achieves this by catalysing conversion of nomilin to deactylenomilin. Hybrids of such fruit with reduced Nom. deactylase activity include Yuzu, Sudachi & Kabosu.
(2) Limnoate dehydrogenase – an enzyme which is isolated from various bacteria such as Arthrobacter globiformis, Corynebacterium fascians, and Pseudomonas sp.
(3) Regulation of UDP-D-glucose: Limonoid Glucosyltransferase.
Applications Of Limonoids
Humans consume on average about 50 to 75g of limonoid glucosides through juices, pulp and other products even including marmalade. On that basis we are seeing plenty of limonin enter the diet and there are many health benefits in doing so.
At the biochemical levels limonoids generally exert their action by a variety of possible mechanisms which we explore a bit further.
Antioxidants
The limonoids all have antioxidant properties. Limonoids have also been shown to induce glutathione S‐transferase enzyme activity which is important for their role as anti-cancer agents. The enzyme is responsible for detoxifying various agents in animals. These activities have been reviewed by Yu et al., (2005) and in fruits like citron and blood oranges (Jayaprakasha & Patil, 2007). This property is related to a host of health benefits.
Another related property is the inhibition of cytochrome P450 oxidoreductase enzymes (CYPs) during metabolic and detoxification processes (Doostdar et al., 2000; Lam & Hasegawa, 1989). This type of inhibitory activity is directly related to their anticancer properties (Poulose et al., 2007).
Anti-Inflammatory & Analgesia
Various limonin derivatives have been synthesized by substituting tertiary amines onto the C-7 position of limonin. One derivative was found to have stronger analgesic activity than aspirin and better anti-inflammatory activity than naproxen (Wang et al., 2018).
Limonin can down regulate the gene for producing nitric oxide synthase and cyclooxygenase-2 which are regulated in part by NF-kB in a rat mucosal cancer model (Vanamala et al., 2006). Building on these studies, a supplement of curcumin and linomin is claimed to modulate particular cell systems involved in regulating inflammation (Kim et al., 2009). Chronic inflammation is a risk factor for certain types of cancers too.
Limonin is also exerts an anti-inflammatory effect in a chronic rheumatoid arthritis model. Limonin was complexed in beta- and gamma-cyclodextrin with high efficiency. This complex was administered in orange juice and was claimed to reduce oedema (da Silva et al., 2018). It is worth noting though that other compounds in the orange juice may well have an important effect.
One of the few human studies found that a citrus extract containing limonin glucoside could reduce certain markers of inflammation by 40%. This was based a double-blind placebo-controlled study in overweight and obese people.
Clinical Studies
Researchers at the Western Human Nutrition Research Center, Agricultural Research Service in the US Department of Agriculture and Department of Nutrition at the University of California in Davis found that the citrus extract decreased particular liver enzymes which are elevated in various chronic diseases such as diabetes, metabolic syndrome or cardiovascular disease (Kelley et al., 2015). The outcome has led to a patent covering the use of limonin glucoside. This patent was prepared for the USDA by Darshan Kelley, lead author on the study.
In the study, ten overweight or obese men and women took part in a cross-over study. They were given orange flavoured drinks formulated with or without limonin for 56 days. The limonin content was 500 mg/day of limonin glucoside which is said to be equivalent to six 8oz glasses of orange juice. The subjects then switched to the other other drink for the next 56 days.
The results demonstrated that limonin glucosides did not affect levels of blood lipids, any lipoproteins, levels of glucose or insulin. Limonin glucoside did however decrease markers of inflammation including MMP-9 and TNF-alpha by 38.7% and 10.7% respectively. The protein MMP-9 is an atherosclerotic plaque destabilisation factor. They also noticed some specific liver based enzymes decreasing in amount – gamma-glutamyl transferase, alkaline phosphatase and alanine aminotransferase. The decreases were 33.8%, 10.1% and 13.1% respectively. These decreases are positive attributes in managing raised levels of inflammation associated with various chronic diseases including liver cancer. the prospect is that limonin glucoside might prevent or even reverse some of these chronic diseases.
A subsequent article from the same group found that the behaviour of various cells associated with managing inflammation did not alter. Limonoid glucoside supplementation did not alter monocyte or T-cell proliferation which indicates there is no site of action with this particular aspect of inflammation.
Antibacterial & Antifungal Activity
Limonoids have some antibacterial and antifungal activity. The limonoids extracted from grapefruit seed such as obacunone seem to be the most potent (Vikram et al., 2010). It along with others can interfere with cell to cell signalling between bacteria like Vibrio harveyi and Escherichia coli O157:H7. These are two examples of food safety organisms that have devastating consequences if they take hold in humans. The furan ring and any C-7 substitution is an important structural feature which upsets these bacteria enough to the extent they cannot form biofilms and can even prevent their adhesion to vegetable leaves and latter parts of the intestine (Vikram et al., 2012).
Anticancer Activity Of Limonoids
There is considerable interest in the compound because it can reduce proliferation of cancer cells, especially colon cancer cells (Jayaprakasha et al., 2009; Patil et al., 2009; Mandadi et al., 2009) and even cause their death as was seen with a particular colon cancer cell line (Chidambara Murthy et al., 2011). Subsequently, limonin has an inhibitory effect against two human cancer cell lines based on the MTT assay in a dose-dependent manner. The cell lines were SNU 449 (liver cancer) and HCT-15 (colon cancer). Interestingly, limonin produced changes in the cancer cell lines but not normal dermal fibroblast cells which suggests it is active against cell which have altered morphology. Various changes to the genetic regulation of apoptosis was noted in these cancer cell lines because of positive alterations in production of apoptosis related proteins (Rahman et al., 2015).
Limonin can induce cell apoptosis and inhibit glycolysis by blocking hexokinase-2 phosphorylation in hepatocarcinoma cancer cell lines (Yao et al., 2018). Some studies with limonin and the same liver cancer cell line suggest it might alter certain types of cell signalling (Tang et al., 2019). It is thought that limonin might be activating PI3K/Akt signaling. One other study indicates that limonin can also supress Wnt signaling in hepatocarcinoma cells.
Limonoids generally induce apoptosis in several cancer cell lines but can also inhibit azoxymethane-induced colon cancer in rats. In one study limonin was shown to inhibit cancer formation (carcinogenesis) of the lung, stomach, buccal pouch and skin in rats (Miller et al., 2004).
The limonoids also have antimutagenic activity according to the Ames test. In one study limonin was antimutagenic against mitomycinC and PhIP in the micronucleus test in vivo (Matsumoto et al., 2017).
A study in mice has demonstrated limonin can suppress intestinal polyp development (Shimizu et al., 2012). More specifically polyp development in the intestines of Apc-mutant mice is also inhibited (Shimizu et al., 2015).
The limonoids generally may be successful treatments for all sorts of intestinal cancers.
Early studies showed the limonoids could diferentially inhibit human cancer cell proliferation as with estrogen receptor-negative and-positive human breast cancer cells in culture (Tian et al., 2001). There is inhibitory activity against human breast cell lines (Kim et al., 2013).
Limonin is also shown to have inhibitory activity against cervical carcinoma cells (Zhao et al., 2019).
Antiviral Activity Associated With Limonoids
Another interesting aspect is that it inhibits HIV-1 replication in cultured macrophages, monocytes and mononuclear cells (Battinelli et al., 2003). It can also inhibit the replication of retroviruses such as HTLV-1. The mode of action could be by inhibition of HIV-1 protease activity.
Anti-Parasitic Activity Of Limonoids
Some recent studies with limonin suggest it could actively prevent the development of the parasite Schistosoma mansoni which is responsible for schistosomiasis. The studies were conducted with infected mice (Eraky et al., 2016).
Auto-Immune Diseases
The use of limonoids generally has been patented as treatment agents against various autoimmune disease (Liu et al., 2017).
Obesity and Hyperglycemia
Metabolic diseases including diabetes have risen in incidence over the last century. One of the target proteins for control is the G protein-coupled bile acid receptor 1 (TGR5). Activation at this receptor prevents obesity and hyperglycaemia (elevated blood glucose levels). Nomilin activates TGR5 in mice which have lived on a high-fat diet (Ono et al., 2011).
One study has shown alleviation of blood pressure (hypertension) in mice. In this example, limonin prevented the MetS induced exaggerated macro- and micro-vascular contractility to phenylephrine (Hassan et al., 2018).
Limonin appears to have some activity against nonbacterial prostatitis (Zhang et al., 2015).
Antifeedant Activity Of Limonoids
On a slightly different tack, the limonoids are effective antifeedants. This means that when they are ingested they prevent insects from example deriving much if any of the benefit they were expecting from chewing through someone’s crop. A number of studies have looked at the way limonin for example stops feeding in larvae or beetle instars for example.
Potatoes are a good example and activity against the Colorado Potato beetle larvae [Leptinotarsa decemlineata (Say)] is one popular study (Alford et al., 1987). Subsequent structure-activity studies have revealed that epoxide and furan groups are important features of molecules that halt feeding by the larvae.
A derivative of limonin which is a 7-methoxime variant is active against Spodoptera frugiperda (Fall Armyworm).
Obacunone and nomilin are very effective antifeedants against termites such as Reticulitermes speratus Kolbe (Serit et al., 1992).
References
Alford, R. A., Cullen, J. A., Storch, R. H., & Bentley, M. D. (1987). Antifeedant activity of limonin against the Colorado potato beetle (Coleoptera: Chrysomelidae). Journal of Economic Entomology, 80(3), pp. 575-578 (Article)
Battinelli, L., Mengoni, F., Lichtner, M., Mazzanti, G., Saija, A., Mastroianni, C. M., & Vullo, V. (2003). Effect of limonin and nomilin on HIV-1 replication on infected human mononuclear cells. Planta Medica, 69(10), pp. 910-913.
Bilal, H., Akram, W., Ali-Hassan, S. (2012) Larvicidal activity of citrus limonoids against Aedes albopictus larvae.
J. Arthropod Borne Dis. 6, pp. 104–111
Champagne, D. E., Koul, O., Isman, M. B., Scudder, G. G., & Towers, G. N. (1992). Biological activity of limonoids from the Rutales. Phytochemistry, 31(2), pp. 377-394
Chandler, B.V., Kefford, J.F., Ziemalis, G. (1968) Removal of limonin from bitter orange juice J. Sci. Food. Agric. 19(2) February pp. 83-86 (Article)
Chandler, B.V. and Johnson, R.L. (1977) Cellulose acetate as a selective sorbent for limonin in orange juice, Journal of the Science of Food and Agriculture, 28, (10), pp. 875-884 (Article)
Chidambara Murthy, K. N., Jayaprakasha, G. K., Kumar, V., Rathore, K. S., & Patil, B. S. (2011). Citrus limonin and its glucoside inhibit colon adenocarcinoma cell proliferation through apoptosis. Journal of Agricultural and Food Chemistry, 59(6), pp. 2314-2323.
Chidambara Murthy, K.N., Jayaprakasha, G.K., Patil, B.S. (2013) Citrus limonoids and curcumin additively inhibit human colon cancer cells. Food Funct. 4(5) pp. 803–810
da Silva, S. A. V., Clemente, A., Rocha, J., Direito, R., Marques, H. C., Sepodes, B., … & Ribeiro, M. H. (2018). Anti-inflammatory effect of limonin from cyclodextrin (un) processed orange juices in in vivo acute inflammation and chronic rheumatoid arthritis models. Journal of Functional Foods, 49, pp. 146-153.
Dandekar, D. V., Jayaprakasha, G. K., & Patil, B. S. (2008). Hydrotropic extraction of bioactive limonin from sour orange (Citrusaurantium L.) seeds. Food Chemistry, 109(3), pp. 515-520. (Article)
Doostdar, H., Burke, M.D. and Mayer, R.T (2000) Bioflavonoids: selective substrates and inhibitors of CYP 4501A1 and CYP1B1. Toxicology 144 pp. 31–38
Eraky, M. A., El-Kholy, A. A. E. M., Rashed, G. A. E. R., Hammam, O. A., Moharam, A. F., Abou-Ouf, E. A. R., … & Hamdan, D. I. (2016). Dose-response relationship in Schistosoma mansoni juvenile and adult stages following limonin treatment in experimentally infected mice. Parasitology Research, 115(10), pp. 4045-4054
Fisher J.F. (1978) An improved method for the quantitation
of limonin in citrus juice by high-pressure liquid chromatography. Journal of Agricultural and Food Chemistry, 26 pp. 497–499 .
Flavian, S. & Levi, A. (1970) A study of the natural disappearance of the limo monolactone in the peel of Shamouti oranges. Inst. J. Food Sci. Technol. 5(2) (Article)
Fong, C.H., Hasegawa, S., Herman, Z., Ou, P. (1989) Limonoid glucosides in commercial citrus juices. J. Food Sci., (Article)
Gualdani, R., Cavalluzzi, M.M., Lentini, G. Habtemariam, S. (2016) TheChemistry and Pharmacology of Citrus Limonoids. Molecules 26(11), 1530 (Article)
Hafeez, F.; Akram, W.; Shaalan, E.A. (2011) Mosquito larvicidal activity of citrus limonoids against Aedes albopictus. Parasitol. Res. 109, pp. 221–229
Hasegawa S., Bennett R.D., Verdon C.P. (1980) Limonoids in citrus seeds: origin and relative concentration. Journal of Agricultural and Food Chemistry, 28 pp. 922–925
Hasegawa, S., Dillberger, A.M., Choi, G.Y. (1984) metabolism of Limonoids: Conversion of Nomilin to Obacunone in Corynebacterium fascians. J. Agric. Food Chem. 32, (3), pp. 457-459 (Article)
Hasegawa, S., & Miyake, M. (1996). Biochemistry and biological functions of citrus limonoids. Food Reviews International, 12(4), pp. 413-435 (Article)
Hasegawa, S., Patel, M. N., & Snyder, R. C. (1982). Reduction of limonin bitterness in navel orange juice serum with bacterial cells immobilized in acrylamide gel. Journal of Agricultural and Food Chemistry, 30(3), pp. 509-511 (Article)
Hasegawa, S., & Pelton, V. A. (1983). Reduction of nomilin bitterness in citrus juices and juice serum with Arthrobacter globiformis cells immobilized in acrylamide gel. Journal of Agricultural and Food Chemistry, 31(1), pp. 178-180.
Hasegawa, S., Vandercook, C. E., Choi, G. Y., Herman, Z., & Ou, P. (1985). Limonoid debittering of citrus juice sera by immobilized cells of Corynebacterium fascians. Journal of Food Science, 50(2), pp. 330-332
Hassan, N. A., El Bassossy, H. M., Fahmy, A., & Mahmoud, M. F. (2018). Limonin alleviates macro-and micro-vascular complications of metabolic syndrome in rats: A comparative study with azelnidipine. Phytomedicine, 43, pp. 92-102 (Article)
Jayaprakasha, G. K., Jadegoud, Y., Nagana Gowda, G. A., & Patil, B. S. (2009). Bioactive compounds from sour orange inhibit colon cancer cell proliferation and induce cell cycle arrest. Journal of Agricultural and Food Chemistry, 58(1), pp. 180-186
Jayaprakasha, G. K., & Patil, B. S. (2007). In vitro evaluation of the antioxidant activities in fruit extracts from citron and blood orange. Food Chemistry, 101(1), pp. 410-418
Kelley, D. S., Adkins, Y. C., Zunino, S. J., Woodhouse, L. R., Bonnel, E. L., Breksa III, A. P., … & Mackey, B. E. (2015). Citrus limonin glucoside supplementation decreased biomarkers of liver disease and inflammation in overweight human adults. Journal of Functional Foods, 12, pp. 271-281 (Article).
Kim, W., Fan, Y.Y., Smith, R., Patil, B., Jayaprakasha, G.K., McMurray, D.N., Chapkin, R.S. (2009) Dietary curcumin and limonin suppress CD4+ T-cell proliferation and interleukin-2 production in mice. J. Nutr. 139, pp. 1042–1048.
Kim, J., Jayaprakasha, G.K., Patil, B.S. (2013) Limonoids and their anti-proliferative and anti-aromatase properties in human breast cancer cells. Food Funct. 4(2) pp. 258–265.
Kola O., Kaya C., Duran H., Altan A. (2010) Removal of limonin bitterness by treatment of ion exchange and adsorbent resins. Food Science and Biotechnology, 19: pp. 411–416 .
Konno, A., Misaki, M., Toda, J., Wadaand, T., Yasumatsu, K. (1982) Bitterness reduction of naringin and limonin by β-cyclodextrin. Agric. Biol. Chem. 46 pp. 2203–8
Lam, L.K.T., Li, Y., Hasegawa, S. (1989) Effects of citrus limonoids on glutathione S-transferase in mice. J. Agric. Food Chem. 37 pp. 878–880 (1989)
Langeswaran, K., Gowthamkumar, S., Vijayaprakash, S., Revathy, R., & Balasubramanian, M. P. (2013). Influence of limonin on Wnt signalling molecule in HepG2 cell lines. Journal of Natural Science, Biology, and Medicine, 4(1), pp. 126.
Liu, L., Li, T., Zhu, G., & Yang, F. (2018). U.S. Patent No. 9,901,566. Washington, DC: U.S. Patent and Trademark Office.
Liu, C., Liu, J., Rong, Y., Liang, N., & Rong, L. (2012). Aqueous extraction of limonin from Citrus reticulate Blanco. Czech Journal of Food Sciences, 30(4), pp. 364-368.
Mandadi, G.K. Jayaprakasha, N.G. Bhat, B.S. Patil, Z. Nature, 62 (2007) 179-188.
Mandadi, M. Ramirez, G.K. Jayaprakasha, B. Faraji, M. Lihono, F. Deyhim, B.S. Patil, Phytomed., 16 (2009) pp. 513-520.
Manlan, M., Matthews, R. F., Rouseff, R. L., Littell, R. C., Marshall, M. R., Moye, H. A., & Teixeira, A. A. (1990). Evaluation of the properties of polystyrene divinylbenzene adsorbents for debittering grapefruit juice. Journal of Food Science, 55(2), pp. 440-445
Manners, G. D. (2007). Citrus limonoids: analysis, bioactivity, and biomedical prospects. Journal of Agricultural and Food Chemistry, 55(21), pp. 8285-8294.
Marina Russo, Adriana Arigò, Maria Luisa Calabrò, Sara Farnetti, Luigi Mondello and Paola Dugo, (2016) Bergamot ( Citrus bergamia Risso ) as a source of nutraceuticals: Limonoids and flavonoids, Journal of Functional Foods, 20, (10-19) (Article)
Matsumoto, T., Takahashi, K., Kanayama, S., Nakano, Y., Imai, H., Kibi, M., … & Watanabe, T. (2017). Structures of antimutagenic constituents in the peels of Citrus limon. Journal of Natural Medicines, 71(4), pp. 735-744.
Miller, E.G.; Porter, J.L.; Binnie, W.H.; Guo, I.Y.; Hasegawa, S. (2004) Further studies on the anticancer activity of citrus limonoids. J. Agric. Food Chem., 52, pp. 4908-4912.
Mishra, P., & Kar, R. (2003). Treatment of grapefruit juice for bitterness removal by Amberlite IR 120 and Amberlite IR 400 and alginate entrapped naringinase enzyme. Journal of Food Science, 68(4), pp. 1229-1233.
Ohta, H. & Hasegawa, S. (1995) Limonoids in Pummelos [Citrus grandis (L.) Osbeck]. J. Food Sci., 60(6) pp. 1284-1285 (Article)
Ono, E., Inoue, J., Hashidume, T., Shimizu, M., Sato, R. (2011) Anti-obesity and anti-hyperglycemic effects of the dietary citrus limonoid nomilin in mice fed a high-fat diet. Biochem. Biophys. Res. Commun. 410, pp. 677–681 (Article)
Patil, R., G. K. Jayaprakasha, K. N. Chidambara Murthy, S. E. Tichy, M. B. Chetti, B. S. Patil. (2009) Food Chem., 114 pp. 1351-1358
Poulose, S. M., Jayaprakasha, G. K., Mayer, R. T., Girennavar, B., & Patil, B. S. (2007). Purification of citrus limonoids and their differential inhibitory effects on human cytochrome P450 enzymes. Journal of the Science of Food and Agriculture, 87(9), pp. 1699-1709.
Qin, S., Lv, C., Wang, Q., Zheng, Z., Sun, X., Tang, M., & Deng, F. (2018). Extraction, identification, and antioxidant property evaluation of limonin from pummelo seeds. Animal Nutrition, 4(3), pp. 281-287 (Article)
Rahman, A., Alam Siddiqui, S., Jakhar, R., & Chul Kang, S. (2015). Growth inhibition of various human cancer cell lines by imperatorin and limonin from Poncirus trifoliata Rafin. seeds. Anti-Cancer Agents in Medicinal Chemistry (Formerly Current Medicinal Chemistry-Anti-Cancer Agents), 15(2), pp. 236-241
Rizza, G. (2016). Citrus limonoids: Functional chemicals in agriculture and foods.
Rouseff, R.L. & Fisher, J.F. (1980) determination of limonin and related limonoids in citrus juice by high performance liquid chromatography. Anal. Chem. 52(8) pp. 1228-1233 (Article)
Roy, A., & Saraf, S. (2006). Limonoids: overview of significant bioactive triterpenes distributed in plants kingdom. Biological and Pharmaceutical Bulletin, 29(2), pp. 191-201.
Shimizu, S., Miyamoto, S., Fujii, G., Nakanishi, R., Onuma, W., Ozaki, Y., Fujimoto, K., Yano, T., Mutoh, M. (2015) Suppression of intestinal carcinogenesis in Apc-mutant mice by limonin. J. Clin. Biochem. Nutr. 57, pp. 39–43 (Article)
Tang, Z., Tang, Y., Li, L., Liu, T., & Yang, J. (2019). Limonin provokes hepatocellular carcinoma cells with stemness entry into cycle via activating PI3K/Akt signaling. Biomedicine & Pharmacotherapy, 117, 109051 (Article).
Tian, Q., Miller, E. G., Ahmad, H., Tang, L., & Patil, B. S. (2001). Differential inhibition of human cancer cell proliferation by citrus limonoids. Nutrition and Cancer, 40(2), pp. 180-184
Todaro, A., Palmeri, R., Scalone, D., Alberio, G. R., Serafini, M., & Spagna, G. (2013). Removal of bitter compounds from citrus byproducts. Italian Journal of Food Science, 25(4), pp. 465.
Tundis, R., Loizzo, M.R., Menichini, F. (2014) An Overview on Chemical Aspects and Potential Health Benefits of Limonoids and Their Derivatives. Crit. Rev. Food Sci. Nutr. 54(2) pp. 225-250 (Article)
Vanamala, J., Leonardi, T., Patil, B.S., Taddeo, S.S., Murphy, M.E., Pike, L.M.,
Chapkin, R.S., Lupton, J.R., Turner, N.D. (2006) Suppression of colon carcinogenesis by bioactive compounds in grapefruit. Carcinogenesis. 27 pp. 1257–65
Vikram, A., Jayaprakasha, G. K., Jesudhasan, P. R., Pillai, S. D., & Patil, B. S. (2012). Limonin 7-methoxime interferes with Escherichia coli biofilm formation and attachment in type 1 pili and antigen 43 dependent manner. Food control, 26(2), pp. 427-438 (Article)
Vikram, A., Jesudhasan, P. R., Jayaprakasha, G. K., Pillai, B. S., & Patil, B. S. (2010). Grapefruit bioactive limonoids modulate E. coli O157: H7 TTSS and biofilm. International Journal of Food Microbiology, 140(2-3), pp. 109-116. https://doi.org/10.1016/j.ijfoodmicro.2010.04.012
Wang., S.-C., Yang., Y., Liu, J., Jiang, A. D., Zhao-Xing, C. H. U., Si-Ying, Chen, Y., … & Zhu, Q.-H. (2018). Discovery of novel limonin derivatives as potent anti-inflammatory and analgesic agents. Chinese journal of natural medicines, 16(3), pp. 231-240. (Article) .
Wilson III, C. W., Wagner Jr, C. J., & Shaw, P. E. (1989). Reduction of bitter components in grapefruit and navel orange juices with beta-cyclodextrin polymers or XAD resins in a fluidized bed process. Journal of Agricultural and Food Chemistry, 37(1), pp. 14-18
Yao, J., Liu, J., & Zhao, W. (2018). By blocking hexokinase-2 phosphorylation, limonin suppresses tumor glycolysis and induces cell apoptosis in hepatocellular carcinoma. OncoTargets and Therapy, 11, 3793.
YT, D. (2011). The advances in the limonoid chemistry of the Meliaceae family. Current Organic Chemistry, 15(9), pp. 1363-1391.
Yu, J., Wang, L., Walzem, R. L., Miller, E. G., Pike, L. M., & Patil, B. S. (2005). Antioxidant activity of citrus limonoids, flavonoids, and coumarins. Journal of Agricultural and Food Chemistry, 53(6), pp. 2009-2014
Zhang, A., Wang, H., Sun, H., Zhang, Y., An, N., Yan, G., … & Wang, X. (2015). Metabolomics strategy reveals therapeutical assessment of limonin on nonbacterial prostatitis. Food & Function, 6(11), pp. 3540-3549.
Zhao, W., Wu, M., Cui, L., & Du, W. (2019). Limonin attenuates the stemness of cervical carcinoma cells by promoting YAP nuclear-cytoplasmic translocation. Food and Chemical Toxicology, 125, pp. 621-628.
Zoccali, M., Arigò, A., Russo, M., Salafia, F., Dugo, P., Mondello, L. (2018) Characterization of Limonoids in Citrus Essential Oils by Means of Supercritical Fluid Chromatography Tandem Mass Spectrometry. Food Analytical Methods 11 pp. 3257 – 3266 (Article)
Zunino, S. J., Storms, D. H., Freytag, T. L., Adkins, Y. C., Bonnel, E. L., Woodhouse, L. R., … & Kelley, D. S. (2016). Dietary supplementation with purified citrus limonin glucoside does not alter ex vivo functions of circulating T lymphocytes or monocytes in overweight/obese human adults. Nutrition Research, 36(1), pp. 24-30 (Article)

Leave a Reply