Plasmids are one of the most important factors in the success of biotechnology.
The word plasmid was introduced by Joshua Lederberg in 1952 as the American molecular biologist who was awarded the Nobel Prize in Physiology /Medicine in 1958. He was just 33 years old but he discovered bacteria could exchange genes when they mated. That prize was shared with Edward L. Tatum and George Beadle for their work on genetics.
A Plasmid: What Is It?
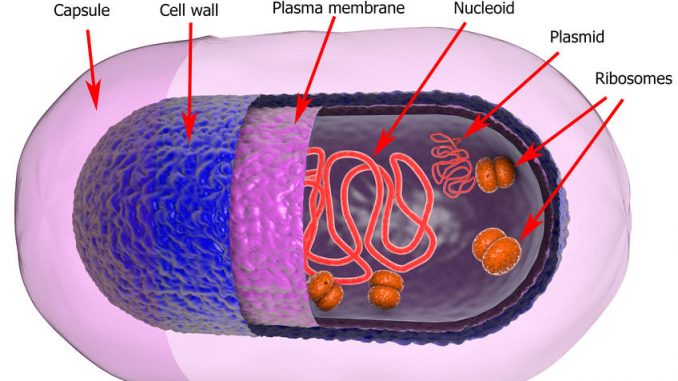
A plasmid is a simple piece of DNA that is separate from chromosomal DNA. It also replicates independently from chromosomal DNA. It is often defined and described as an autonomously replicating piece of DNA.
Plasmids are double stranded and in most cases circular.
Plasmids occur naturally in bacteria but are sometimes found in eukaryotic organisms. Consider the 2-micrometer-ring in the brewing yeast, Saccharomyces cerevisiae. It is thought that they evolved in bacteria/prokaryotes and were part of their make-up. They are best known for expressing products from antibiotic resistance genes which also means they develop resistance to antibiotics.
The value of a plasmid is it can be modified by carrying a piece of inserted DNA which is inserted artificially. These vehicles are critical in synthetic biology. It will also replicate in E.coli bacteria which means the bacteria now becomes a plasmid DNA factory. It is possible to insert what is desired as a gene into the plasmid and have it replicate sufficient amounts of a desired protein without destroying the insert during the process. For the biotechnologist, they can now express proteins from all sorts of sources.
Plasmid Sizes
A bacteria has at least one circular chromosome which is often of 4.3 million base pairs (b.p.) with a length of 1.3mm. A plasmid is only 5,000 base pairs but there can be many of them residing in a cell. They only carry a few genes.
Multiplication Of Plasmids
They have a single origin of replication.
They are replicated by exactly the same machinery as for bacterial chromosomes. Some plasmids are copied at the same rate as a typical chromosome so a single cell often only has a single copy of a plasmid. Other plasmids are copied at a higher rate and can carry over 50 or more of them.
Plasmids can enter a bacteria with considerable ease. It probably accounts for there ability to confer antibiotic resistance in bacteria. That property also makes bacteria ideal as host cells because they can be transformed so easily with a recombinant piece of DNA.
Plasmids Can be Classified On the Basis Of Function
Plasmids can be classified further into two basic groups – conjugative and non-conjugative. The conjugative plasmids mediate their own transfer between bacteria using the process of conjugation. This requires functions specified by the tra (transfer) and mob (mobilising) regions that are carried n the plasmid. Plasmids most often described as conjugation plasmids are the fertility plasmids, usually called F Plasmid. These carry fertility genes (i.e. tra-genes) needed for conjugation and allow the transfer of genetic information between two cells.
The non-conjugative plasmids are not self-transmissable. They may be mobilised by a conjugation-capable plasmid if their mob region is operational.
We can also classify plasmids based on the number of copies of the plasmid found in the host cell. This is the copy number. Plasmids with a low copy number in the host bacteria show stringent control of DNA replication where replication of the pDNA (plasmid DNA) is closely tied to the host cell’s chromosomal DNA replication. The high copy number plasmids are described as relaxed plasmids because DNA replication is not dependent on chromosomal DNA replication of the host.
Generally, conjugative plasmids such as the F plasmid are large, have stringent control of DNA replication and are present in low copy numbers. The non-conjugative plasmids are much smaller, show relaxed DNA replication and are found in host cells with high copy numbers.
Naturally occurring plasmids have been modified further to make them suitable for biotechnology applications. We mentioned the Resistance plasmids (R Plasmid) that contains genes for building machinery for dealing with poisons and antibiotics. The Col Plasmids are those that encode the antibacterial polypeptides called bacteriocins which is a protein that kills other strains of bacteria. The col proteins of E. coli are encoded by genes such as Col E1. ColE1 is one of the smallest plasmids being only 7 kilobases. It is non-conjugative and produces between 10 and 15 copy numbers. .
Naming of Plasmids
The nomenclature for a plasmid cloning vector is well established. Take pBR322 which conforms to standard rules for vector nomenclature. The ‘p’ indicates it is a plasmid. The ‘BR’ establishes the laboratory in which the vector was originally constructed. In this example, BR stands for Bolivar and Rodriguez who the two researchers who invented the plasmid. The ‘322’ is the unique number of plasmid developed in this particular laboratory. Others are pBR325 etc.
First uses In Biotechnology
Plasmids are the first type of cloning vector where they are used to clone DNA in bacteria. These engineered plasmids are used to clone genetic material of up to 10,000 base pairs.
The Top Plasmids to Use
pBR322
pBR322 is one of the first widely used plasmids in molecular biology. It carries genes for resistance to ampicillin and tetracycline, enabling selection of transformed cells with these antibiotic markers. pBR322 has a variety of restriction sites, making it versatile for DNA cloning and manipulation.
The first and perhaps most important feature is the size of this plasmid. Cloning vectors should be less than 10 kb because it avoids issues such as DNA breakdown during purification.
The pBR322 plasmid has also what are now classic features. It is 4,363 base pairs in length. It means the vector can be purified with ease as well as the recombinant DNA molecules constructed from it. Even if you add an extra 6kb of additional DNA, a recombinant pBR322 is of a suitable size for purification.
It has a single origin of replication (ori) within a structural element of the DNA called the replicon. We might also see it written on specifications as the replicon rep because it is responsible for the replication of the plasmid.
It contains two genes that confer resistance to two different antibiotics – for ampicillin (amp) and for tetracyclin (tet). These are selectable markers for any cell containing the plasmid. each marker gene includes unique restriction sites that can be used in cloning.
Insertion of new DNA into pBR322 that has been restricted with Psti, Pvul or Scal will inactivate the ampR gene. Insertion using any one of the 8 restriction endonucleases, notably BamH1 and HindIII inactivates tetracycline resistance.
With the large number of restriction sites, these can be used for insertional inactivation which mans that pBR322 can be used to clone DNA fragments with any of the sticky ends.
The other benefit is that this plasmid has a reasonably high copy number. Generally, there are 15 molecules present in a transformed E. coli cell but the number can be increased to 1000 to 3,000 by plasmid amplification in the presence of a protein synthesis inhibitor such as chloramphenicol. On this basis, culturing E.coli produces a good yield of recombinant pBR322 molecules.
pUC19
The pUC19 plasmid is a widely used cloning vector derived from the pUC series of plasmids. It carries the ampicillin resistance gene, allowing for selection of transformed bacterial cells. It also contains multiple cloning sites for inserting DNA fragments and is commonly used for routine cloning and DNA amplification.
pET vector series
The pET vector series, such as pET-28a and pET-21a, are commonly used for recombinant protein expression in Escherichia coli (E. coli). These plasmids have a strong T7 promoter for inducible protein expression, along with antibiotic resistance markers. They also include a His-tag for purification and other useful features for protein production.
pGEX vector series
The pGEX vector series, including pGEX-4T-1 and pGEX-6P-1, are widely used for the expression and purification of fusion proteins using glutathione S-transferase (GST) affinity chromatography. These plasmids allow for fusion of the target protein with GST, facilitating purification and downstream applications.
pBlueScript
pBlueScript is a versatile cloning vector commonly used in molecular biology research. It carries the ampicillin resistance gene and contains multiple cloning sites. pBlueScript is derived from the pUC series and is particularly useful for subcloning DNA fragments and generating plasmid DNA templates for sequencing.
pBluescript SK+
pBluescript SK+ is a cloning vector derived from pBluescript II. It carries the ampicillin resistance gene and features a multiple cloning site. Additionally, it contains sites for in vitro transcription and translation, making it useful for in vitro expression and mutagenesis studies.
pCMV vector series
The pCMV vector series, such as pCMV6-Entry and pCMV-Tag2B, are popular expression vectors for mammalian cell transfection. These plasmids contain the cytomegalovirus (CMV) promoter for strong constitutive expression of the inserted gene. They also include antibiotic resistance markers for selection in mammalian cells.
pEGFP-N1
pEGFP-N1 is a widely used plasmid for expressing green fluorescent protein (GFP) in cells. It contains the CMV promoter and the enhanced GFP gene, allowing for visualization of transfected cells under fluorescence microscopy. pEGFP-N1 also carries a neomycin resistance gene for selection in mammalian cells.
pACYC184
pACYC184 is a low-copy-number plasmid commonly used in genetic engineering. It carries the chloramphenicol resistance gene and provides a stable replication system. pACYC184 is useful for maintaining and propagating large DNA inserts, carrying out complementation studies, and for applications requiring low-copy-number plasmids.
pETBlue-2
pETBlue-2 is a versatile cloning vector designed for protein expression and purification in E. coli. It combines features of pET vectors and pBlueScript. It contains a T7 promoter.
The Ti-Plasmid
The Ti-plasmid is in reality one of the most important plasmids available especially in the creation of transgenic plants. This plasmid was first isolated from Agrobacterium tumefaciens which produced crown gall tumors in dicotyledons. A small segment of the plasmid called T-RNA is transferred to and stably maintained in tumour based plant cells. One of its most important applications has been the generation of transgenic sugar beet. Creating transgenic plants has come about because of the commercial desire to establish resistance against plant diseases.
Article revised from 2nd June 2020 with updated knowledge on plasmids.
References
Bevan, M., Barnes, W. M., & Chilton, M. D. (1983). Structure and transcription of the nopaline synthase gene region of T-DNA. Nucleic acids research, 11(2), pp. 369-385.

I’ve been using this article to supplement my notes. I noticed my lecturers were copying stuff from your website so it must be good.