Cannabis and products that contain it have exploded onto the food and consumer healthcare marketplace. At the heart of this particular herb is cannabidiol (CBD). A growing market for wellness products has largely driven the legal use of CBD around the world. Its use has triggered a rethinking of consumer interaction with a product which is still thought to be edgy and a liability in society.
At the present time its use is still heavily regulated but countries have legalized it, not as cannabis but as hemp and products containing hemp. Hemp has a more benign connotation in society because its fibre used in clothing. However it is a more acceptable term for the cannabis plant.
Canada became the first of the G7 nations to legalize cannabis for recreational use through the Cannabis Act and Cannabis Regulations which fit hand in glove with each other. The USA is slowly following suit as they seek to understand the impact of CBD in particular on consumer mood and behaviour.
What Is CBD?
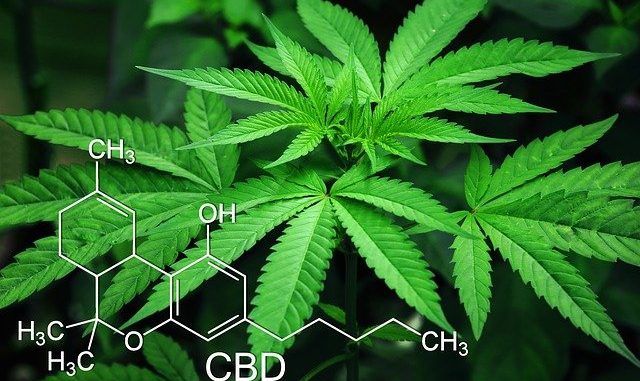
CBD or cannabidiol is one of nearly a hundred cannabinoids found in the cannabis family of plants. It is the most abundant of these types of compounds.
These compounds are secreted by cannabis plants in their trichomes which are at the tips of the flower. This is where the highest concentrations are to be found. These cannabinoids are sometimes called phytocannabinoids.
THC or Δ9-Tetrahydrocannabinol is the psychoactive form that smokers of marijuana would be most familiar with. It is not however the same compound as CBD which is the therapeutic variant.
The plant produces these compounds in an acidic form i.e. a compound with an attached carboxyl group. THC-A (Δ9-Tetrahydrocannabinolic acid) is converted to THC only through the action of heat. This is a decarboxylation reaction and only when this occurs is it converted to the more potent psychoactive form. The same heat reaction must occur to the acidic form of CBD to generate the therapeutic form.
The use of CBD oil and CBD itself has been a pharmaceutical product or healthcare product for a number of years but new legislation really drove CBD use.
In 2018, the USA passed the Farm Bill which legalised the growing of hemp. The bill also enabled hemp-derived products containing less than 0.3% tetrahydrocannabinol (THC) on a dry weight basis to be removed from the illegal drug Controlled Substances List.
Following this ruling, hemp farming increased considerably and with it the enormous development of the CBD industry.
Market analysis according to the Brightfield Group reckons the US market for CBD products at the end of 2019 was estimated at $4 billion. Following the 2018 Farm Bill, the market increase was 562% from the end of 2018 which is an enormous rise.
Marijuana, Hemp And Cannabis
Another important distinction to make is the difference between hemp, marijuana and cannabis. Marijuana is the form of cannabis which is smoked because of its high THC content.
Hemp is the form grown for commercial production of clothing fibres even though they both refer to the same plant species. Hemp is a grain too that has a cultivation history of 10,000 years and has a much lower THC level. The cultivation of hemp has led to much fibrous stalks with a height as great as possible.
Marijuana has been cultivated to maximise the THC content with shorter plants and more leaf density. the overall cannabinoid content is also much higher.
Current US Regulations On CBD
The regulatory situation for CBD in the USA is still a complex and uncertain issue.
The 2018 Farm Bill which is still being revised kick started the legal growing of hemp or cannabis where the THC content was less than 0.3%. This plant was removed from the Controlled Substance Act and has subsequently become an agricultural commodity. In March 2021, the Farm Bill was updated to its final form and can be explored more fully as part of the US Domestic Hemp Production Program on the Youtube web-site. The guidance covers agronomy, growing conditions, processing of the hemp plant and the creation of CBD products. It’s recommended to anyone wishing to understand more fully the product development elements of this material.
The Federal Food, Drug and Cosmetic Act (FFDCA) is the main piece of legislation covering all drugs and foods for sale. One section, Section 201 (ff)(3)(B) states that “a dietary supplement does not include an article authorized for investigation as a new drug, antibiotic, or biological for which substantial clinical investigations have been instituted and for which the existence of such investigations has been made public.”
Section 301(II) of the FFDCA prohibits the introduction or delivery for introduction into interstate commerce of any food (including a dietary supplement) that contains an approved drug or biological product, or a drug or a biological product for which substantial clinical investigations have been instituted and their existence made public.
A good example of such a drug using CBD is Epidiolex which required significant clinical investigation for its use as an epilepsy treatment. Successful completion of the trials meant it was approved for use in the USA.
On this basis, cannabidiol cannot be sold as an ingredient in foods or dietary supplements until new legislation could occur.
Come 2020, a bipartisan bill (H.R 5587) was introduced on the 13th January to the House of Representatives to amend the FFDCA so that CBD was exempt from both Sections 201(ff)(3)(B) and 301(II) of that bill. This bill however was not passed!
In March 2020, the FDA suggested it would be open to creating a pathway for CBD to become regulated as a dietary supplement. It opened up a consultation requesting further information.
The current position: the FDA still contends that it is illegal to add CBD to foods or to label it a dietary supplement. The FDA does not recognize CBD as GRAS (Generally Recognized as Safe) or as a legal food additive. Because of its approval as a drug, CBD is thus still prohibited in foods under the FFDCA.
At this moment in time there are two probably regulatory pathways for CBD in food. It can either be a prospective rule passed by the FDA and through a congressional regulation. The current bill being discussed is the Hemp Access and Consumer Safety Act which is a bill to be introduced in May 2021.
OK then, it is still illegal to market CBD containing products and whilst it is widely available, the FDA regularly issues warning letters to businesses and companies that advertise and sell products especially if they claim it treats conditions like cancer, Alzheimer’s Disease, fibromalgia and other conditions. In other words any unsubstantiated claims must not be made.
The FDA continues to evaluate the long-term safety of CBD where the THC level is less than 0.3%.
CBD And The Situation With The Various States Of The USA.
At the state level, CBD is allowed in food in 22 states at the moment which entirely contradicts the law at the federal level. This fragmented situation is similar for use of cannabis/marijuana in these states. So in Oregon, Colorado, Alaska, New Mexico and a number of mid-west states, CBD can be specifically allowed in food.
In some states like California, Montana, Nevada, Arizona for example there is no permission for adding CBD to food. It’s complicated to say the least!
Where it is permitted, there is considerable growth in those marketplace for these products.
Because of COVID-19, development of CBD containing products has actually slowed considerably but this may well improve as the country comes out of any lockdown measures.
Another trend to follow is the development of delta-8 CBD products although this compound has psychoactive properties in its own right. The issue is that it would complicate the situation further and actually halt regulation of CBD in the USA.
The Position Of CBD In Canada
Canada was one of the first countries to open its doors to cannabis products especially ones with CBD. Back in 2019 there was consultation to allow CBD into some foods.
Health Canada does not discriminate between cannabis and hemp. In fact edible cannabis means ‘products containing cannabis that are intended to be consumed in the same manner as food which means eaten and drunk’.
The provisos however are that they can only be sold in licensed dispensaries. They also have to state the THC content is less than 10mg per packet. They cannot have vitamins, alcohol, caffeine, nicotine or other functional ingredients added to them. They must make no dietary or health claims and they cannot appeal to the young.
The Position of CBD In The European Union
In the European Union, CBD has also posed a number of issues and it has yet to fully resolve the situation over the regulation of this compound especially in food. Interestingly the issues that affect Canada and the USA are different to those in Europe! The differences are down to the way the EU legislates on new foods before they can ever get to market.
Any new type of food is treated as a novel food unless it has a recognizable past history of use. A novel food must go through an approval process. This begins with an application to get the ball rolling for final acceptance.
Any business outside Europe but involved in the CBD industry would find this type of process of approval a very different experience to that in the USA or Canada. A significant number of businesses anyway tried to argue that the use of CBD had a historical past and did not need novel foods approval because it was derived from hemp. Hemp and hemp seed in particular was an agricultural product that had been available for centuries which contradicted any sense of novelty. The EU, through The European Food Standards Agency (EFSA) did not accept the idea of hemp having this past history of traditional food use, insisting it was a novel food. On that basis, the European Commission (EC) using EFSA began to receive applications for CBD based products for novel food approval.
Another source of confusion was the change in attitude of the EC back in June 2020 when it then froze all applications for novel foods because it needed to decide whether CBD derived from hemp flower was actually a narcotic. At the time the United Nations (UN) has and still has strict rules on narcotics. In the November 2020 ruling from the EU’s Court of Justice commonly called the Kanavape ruling, it was decided that CBD from hemp was not a narcotic according to UN convention. After that ruling the novel food applications were resumed but not before it had sown doubt throughout the EU about the safety of CBD.
To heap on further confusion, member countries can still pass their own legislation on particular foods and there has been a shift in recent years to take CBD containing products of all types off the shelf. This applies to CBD in sublingual products as well as foods.
However from December 2020, the novel foods application process for CBD containing foods is still operating openly supported by EFSA with the help of the European Commission (EC). As expected a dossier needs to be submitted containing good quality safety data and toxicology studies to support the application that these foods can enter the supply chain. Most of the applications appear to be synthetic CBD products of one description or another. It remains to be seen whether there is a shift in these types of products but its worth keeping a ‘weather eye’ on these applications.
A key element of any data submitted is that the acute reference dose for THC of 1 microgram per kg of body weight is not exceeded. The amount is extremely low. The issue here is that EU member states have different approaches to THC content.
The Novel Food Process For The UK
The UK’s Food Standards Agency now operates almost independently of the EC following the end of the Brexit provisional period on the 31st December 2020. The UK is seen as a major market for CBD products anyway so there will be a stronger focus here for approval than in the EU.
From what can be seen, all the legislative processes that the UK was part of in the EU have remained in principle going forward. It has now accepted applications from the 1st January 2021 onwards using a parallel UK novel foods process which operates at this moment in time like the EU’s version.
Given the nature of the UK’s lighter touch on bureaucracy, the process may be quicker for novel foods approval generally. CBD products that were available in the marketplace were allowed to stay if the application was validated by the FSA. A deadline for new applications was the 31st March 2021. It will probably be 2022 when any new approvals are granted.
Consumer Attitudes In the United States
Fifteen states in the USA legalized recreational marijuana and over 30 have legalized medical marijuana. Cannabis products including edibles are illegal at the federal level.
A number of surveys have sought to understand the market for CBD products. Intertek Health Sciences examined the attitudes data from the USA using four well-developed surveys which they explained at the IFT’s online conference in 2020. We’ve attempted to summarise them here.
Corroon & Phillips (2018) collected survey data to basically work out how and why individuals were using cannabidiol. This relied on an online survey obtained through social media. It was promoted by herbal vaporizer producers and those marketing CBD products. POLITICO/Harvard TH Chan School of Public Health examine the public’s views in CBD regulations in 2017. This was a solid examination on 1,000 randomly selected adults who were asked their views. Wheeler et al., (2020) examined CBD knowledge, attitudes and cannabis use in young adults. This too was an online survey using social media. The final report from HelloMD and Brightfield Group (HBG, 2017) looked at the experiences of the HelloMD online community. This group were asked about their perceptions and experiences of US medicinal cannabis products.
About 55% of people obtain their CBD from the dispensary, 28% from a local delivery service and the the remainder from online purchase and smaller numbers from shops or by personal growing (HelloMD & Brightfield Group, 2017).
Beliefs On CBD Regulation In The USA
Some of the conclusions from the various surveys show that over half the participants agree that CBD products should be allowed. They should be available to the public in the same manner as dietary and vitamin supplements, without first being declared as safe (56%) or effective (65%) for use by the FDA.
Frequency Of CBD Use
CBD was administered in many different ways (HBG, 2017). Most consumers apparently took CBD using different methods. The most common were vaping and oil cartridges, tinctures, topicals and edibles.
The Corroon & Phillips (2018), Wheeler et al., (2020) and HBG (2017) studies found that most CBD users took it once daily.
There seems to be quite a range in the dosage level from less than 10mg to less than 100mg. It seems from the HBG study that most consumers prefer a CBD dose less than 10mg.
Dosage To Take
The Wheeler et al., (2020) study looked at how consumers estimated or calculated the amount of CBD to use. Thus, 38% of users took a product label recommendation but only 3% relied on the healthcare provider. The remainder (21%) based their use on whether they felt an effect or not.
It seems that CBD users have a poor framework for estimating the dosage levels. In the scientific community, not much is known about the optimal dose although some researchers believe CBD has a bell-shaped dose response curve.
Interestingly, 54% agree that consumers should still be able to buy a CBD product even if the FDA considers the evidence that it is unsafe. However, 74% agree that a business marketing health claims can only do so if it has been scientifically proven.
The Intertek Health Sciences (2020) conclusion considered that CBD users took it for relieving pain, inflammation and anxiety. It was also a sleep aid. There seemed to be more than one route for each consumer and many used a variety of routes of administration. These could be by topical, inhalation and oral routes.
For many users there was confusion about the dosage level and frequency and they were obtaining their information from mostly poor and unreliable sources. There was also a situation where side effects were commonly experienced which were damaging and in some cases highly dangerous.
CBD With Alcohol
One of the most difficult areas to navigate is the combination of CBD with alcohol. The Portman Group in the UK considers that product names and descriptors such as ‘CBD gin’ and ‘rum infused with CBD’ is an issue. Alcohol and CBD is a highly problematic combination.
The references to CBD have to be proved in a factual and ‘non-emotive’ manner. All producers must seek legal advice before any products with CBD especially with alcohol, onto the market.
In the USA and Europe, over 3/4 of consumers believe that health claims must be fully researched and scientifically proven. The general feeling in the CBD user community is that safety and efficacy needs to be comprehensively investigated.


Such insights!
We get a lot of hemp oil through your site! I really like the CBD oil for pets from AMZ Evergreen. Glad you redid the portal for purchase of this product from your website. We can’t it from them directly. Don’t know why but it does seem strange you can only buy it through your site and then onto Amazon.